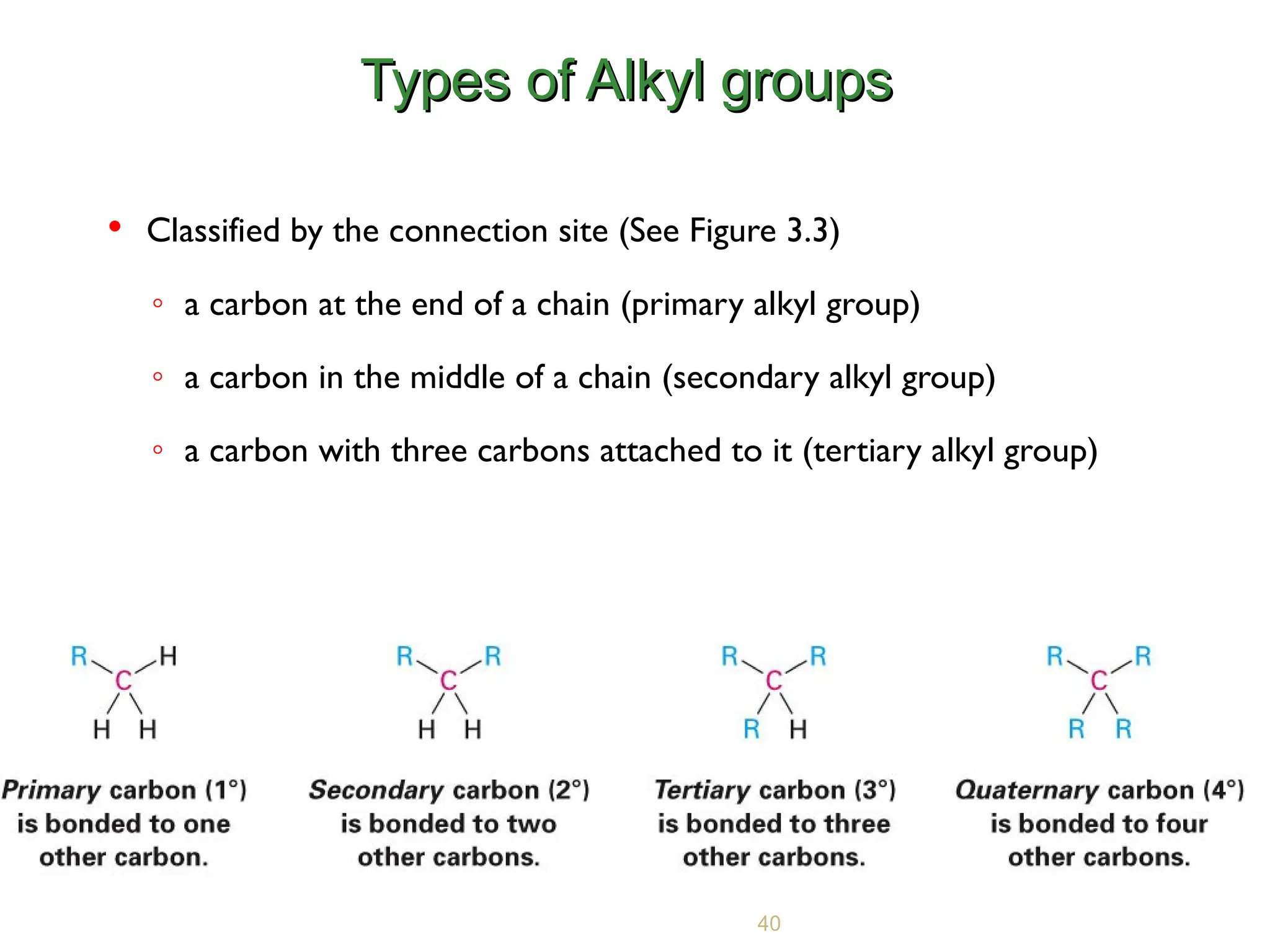
Understanding the structure and properties of organic compounds is fundamental to the study of chemistry. One of the key concepts in organic chemistry is the sorting of carbons base on the act of other carbon atoms they are bind to. This assortment helps in prefigure the reactivity and behavior of organic molecules. The primary, secondary, and tertiary carbons are important in this context, each play a distinct role in the chemic properties of compounds.
Understanding Primary, Secondary, and Tertiary Carbons
In organic chemistry, carbons are assort free-base on the number of other carbon atoms they are directly bonded to. This sorting is essential for understanding the reactivity and constancy of organic compounds. The three principal types of carbons are primary, petty, and tertiary carbons.
Primary Carbons
Primary carbons are those that are bond to only one other carbon atom. These carbons are typically found at the ends of carbon chains or branches. Primary carbons are less sterically hindered, create them more reactive in commutation and elimination reactions. for instance, in the molecule ethane (CH3CH3), both carbon atoms are primary carbons.
Secondary Carbons
Secondary carbons are bonded to two other carbon atoms. These carbons are found in the middle of carbon chains or branches. Secondary carbons are more sterically block than chief carbons, which can impact their reactivity. For example, in propane (CH3CH2CH3), the middle carbon is a secondary carbon.
Tertiary Carbons
Tertiary carbons are bonded to three other carbon atoms. These carbons are typically found at branch points in carbon chains. Tertiary carbons are the most sterically hinder, which can significantly encroachment their reactivity. An example is isobutane ((CH3) 3CH), where the primal carbon is a third carbon.
Importance of Primary, Secondary, and Tertiary Carbons in Organic Chemistry
The assortment of carbons into main, secondary, and third is important for several reasons:
- Reactivity: The reactivity of a carbon atom is influenced by its assortment. Primary carbons are loosely more responsive due to less steric hindrance, while tertiary carbons are less responsive due to greater steric hindrance.
- Stability: The stability of carbons also varies base on their assortment. Tertiary carbons tend to form more stable carbocations due to the inducive effect of the surrounding alkyl groups.
- Stereochemistry: The spacial arrangement of atoms around a carbon can regard the stereochemistry of a molecule. Understanding the sorting of carbons helps in predicting the stereochemical outcomes of reactions.
Examples of Primary, Secondary, and Tertiary Carbons in Organic Compounds
To better realize the concept of master, secondary, and tertiary carbons, let's look at some examples of organic compounds and name the types of carbons stage in each.
Ethane (CH3CH3)
In ethane, both carbon atoms are primary carbons because each is bonded to only one other carbon atom.
Propane (CH3CH2CH3)
In propane, the middle carbon is a junior-grade carbon because it is bonded to two other carbon atoms. The end carbons are principal carbons.
Isobutane ((CH3) 3CH)
In isobutane, the central carbon is a tertiary carbon because it is bonded to three other carbon atoms. The other carbons are primary carbons.
Reactivity of Primary, Secondary, and Tertiary Carbons
The reactivity of primary, secondary, and 3rd carbons is influenced by their steric hitch and the stability of the intermediates formed during reactions. Here's a breakdown of their reactivity:
Substitution Reactions
In permutation reactions, the reactivity of carbons follows the order: master junior-grade tertiary. This is because master carbons are less sterically hindered, make it easier for nucleophiles to attack. Tertiary carbons, being more sterically obstruct, are less reactive in substitution reactions.
Elimination Reactions
In excretion reactions, the reactivity of carbons follows the order: third lower-ranking primary. Tertiary carbons form more stable carbocations, which facilitates the elimination process. Primary carbons, being less stable, are less responsive in excretion reactions.
Stereochemistry and Primary, Secondary, and Tertiary Carbons
The spatial arrangement of atoms around a carbon atom can significantly touch the stereochemistry of a molecule. Understanding the sorting of carbons helps in predicting the stereochemical outcomes of reactions. for representative, the formation of chiral centers in organic compounds often involves the response of primary, secondary, or tertiary carbons.
In chiral molecules, the configuration around a carbon atom can be either R or S, depending on the priority of the substituents. The classification of carbons as main, secondary, or tertiary can help in determining the precedency of substituents and predicting the stereochemical outcome of a reaction.
Applications of Primary, Secondary, and Tertiary Carbons
The sorting of carbons into master, secondary, and 3rd has numerous applications in organic chemistry. Some of the key applications include:
- Synthesis of Organic Compounds: Understanding the reactivity of primary, secondary, and tertiary carbons is crucial for the synthesis of organic compounds. Chemists use this noesis to design man-made routes that maximize yield and selectivity.
- Drug Design: In pharmaceutical chemistry, the assortment of carbons is all-important for design drugs with specific biological activities. The reactivity and stability of carbons can affect the adhere affinity and selectivity of drugs.
- Petrochemical Industry: In the petrochemical industry, the classification of carbons is used to understand the reactivity of hydrocarbons and design catalytic processes for their conversion into useful products.
Note: The sorting of carbons into main, secondary, and 3rd is a fundamental concept in organic chemistry that has across-the-board ranging applications in various fields.
Conclusion
In drumhead, the classification of carbons into primary, secondary, and tertiary is a crucial concept in organic chemistry. Understanding the reactivity, stability, and stereochemistry of these carbons is essential for predicting the demeanor of organic compounds and designing synthetic routes. The applications of this sorting range from the synthesis of organic compounds to drug design and the petrochemical industry. By mastering this concept, chemists can gain deeper insights into the properties and reactions of organic molecules, leading to advancements in diverse fields of chemistry.
Related Terms:
- nitrogen bind to 3 carbons
- methyl main lowly tertiary
- main and 3rd carbon example
- main vs secondary 3rd carbons
- primary secondary tertiary amine
- 3rd secondary master carbocation