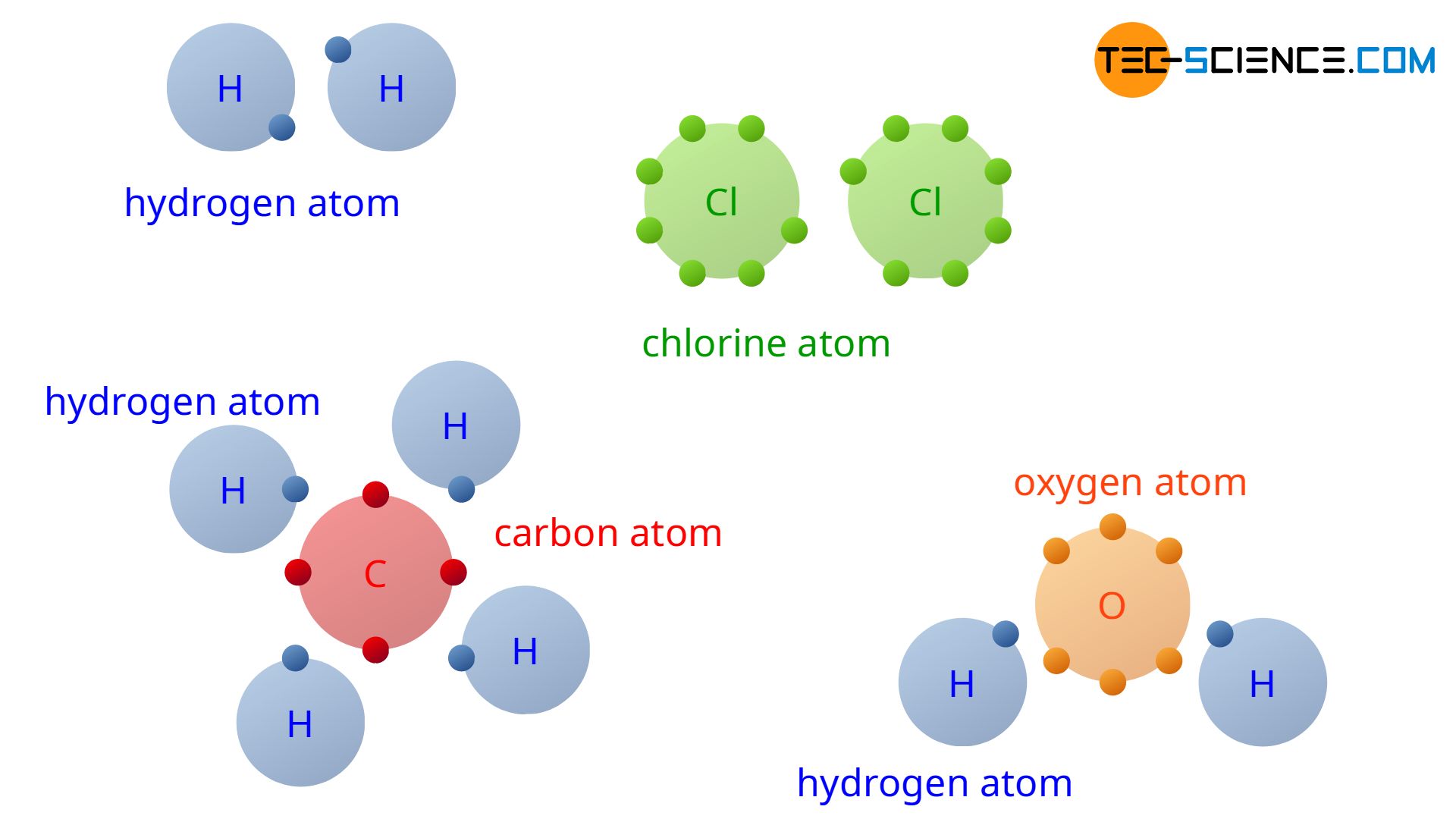
Carbon is one of the most versatile elements in chemistry, forming the backbone of organic life and countless semisynthetic materials. A central enquiry in understand carbon s behaviour is: How many covalent bonds can each carbon atom form? Unlike many other elements, carbon s alone ability to form four strong covalent bonds enables its remarkable content to make diverse molecular structures from simple hydrocarbons to complex biomolecules. This versatility stems from carbon s atomic configuration: with six valency electrons, it achieves constancy by sharing four electrons, organize four tantamount covalent bonds. Whether in methane (CH₄), diamond, or DNA, carbon systematically forms four bonds, making it the foundation of organic chemistry. But how precisely does this bind act, and what limits or exceptions exist? Exploring the construction and bonding patterns reveals why four is the maximum number carbon can sustain under normal conditions. Carbon s electron contour is key to understanding its bonding capability. With six electrons in its outermost shell, carbon seeks to complete its valency bed by share four electrons two pairs through covalent bonds. Each share pair counts as one bond, allowing carbon to bond with up to four different atoms. This tetravalency defines carbon s role in constitute stable molecules across biology, industry, and materials skill. The power to form four bonds explains why carbon forms chains, rings, and three dimensional networks, enabling the complexity seen in proteins, plastics, and minerals.
Understanding Covalent Bond Formation in Carbon Covalent adhere occurs when atoms share electrons to attain a entire outer energy level. For carbon, this operation involves crossing a rearrangement of nuclear orbitals to maximise bonding efficiency. The most mutual cross in organic compounds is sp³, where one s and three p orbitals mix to form four tantamount sp³ hybrid orbitals. Each orbital overlaps with an orbital from another atom, make a potent covalent bond. This hybridization ensures equal bond strength and geometry, typically tetrahedral, which minimizes electron repulsion. The resolution is a stable electron distribution that supports four unmediated connections. The tetrahedral arrangement around carbon allows flexibility in molecular geometry. In methane (CH₄), for illustration, four hydrogen atoms occupy the corners of a tetrahedron, each bonded via a single covalent link. This spacial orientation prevents steric clashes and stabilizes the molecule. Similarly, in ethane (C₂H₆), each carbon forms four bonds three to hydrogen and one to the other carbon demonstrating how carbon balances multiple attachments through directing bonding.
While carbon typically forms four covalent bonds, certain conditions and structural contexts can influence this pattern. In some allotropes and high pressure environments, carbon adopts different adhere geometries, but these remain rare and often unstable under standard conditions. For example, diamond features sp³ hybridized carbon atoms arranged in a rigid 3D lattice, where each carbon shares four bonds but in a fixed tetrahedral net. In contrast, graphene consists of sp² interbreed carbon atoms constitute a flat hexagonal sheet, with three bonds per carbon and one delocalized π electron contribute to exceptional conduction. These variations foreground how interbreeding affects bonding density but do not change the underlying limit of four bonds per carbon atom.
Note: Carbon seldom exceeds four covalent bonds due to its electronic structure; outgo this leads to imbalance or requires extreme conditions.
Another aspect to consider is bond strength and length. The average bond length in a C C single bond is about 154 picometers, while C H bonds are shorter (137 pm). These distances reflect optimal orbital overlap and electron sharing efficiency. When carbon attempts to form more than four bonds, the geometry becomes strained, increasing repulsion between electron pairs and countermine overall stability. This explains why hypervalent carbon compounds those with more than four bonds are uncommon and usually require specialise ligands or metal coordination, such as in certain organometallic complexes.
Note: Carbon s maximum of four covalent bonds ensures molecular stability; outdo this typically results in structural distortion or decomposition.
In drumhead, carbon s ability to form four covalent bonds arises from its electronic form, sp³ hybridization, and tetrahedral geometry. This logical bond pattern underpins the diversity and complexity of organic and inorganic compounds alike. While exceptions exist in specialized chemic environments, the rule remains clear: carbon forms four stable covalent bonds under normal circumstances. This capacity enables the rich chemistry that sustains life and drives innovation across scientific fields. Understanding this fundamental principle helps explain not only introductory molecular behavior but also the design of progress materials and pharmaceuticals rooted in carbon based structures.
Note: The tetrahedral bonding model is essential for predicting molecular shape, reactivity, and physical properties in carbon control systems.