Understanding the freezing point in centigrade is crucial for various scientific, industrial, and everyday applications. The freezing point, ofttimes advert to as the dethaw point when transition from solid to liquid, is a key property of substances. This blog post delves into the significance of the freezing point in centigrade, its measurement, and its applications in different fields.
What is the Freezing Point in Centigrade?
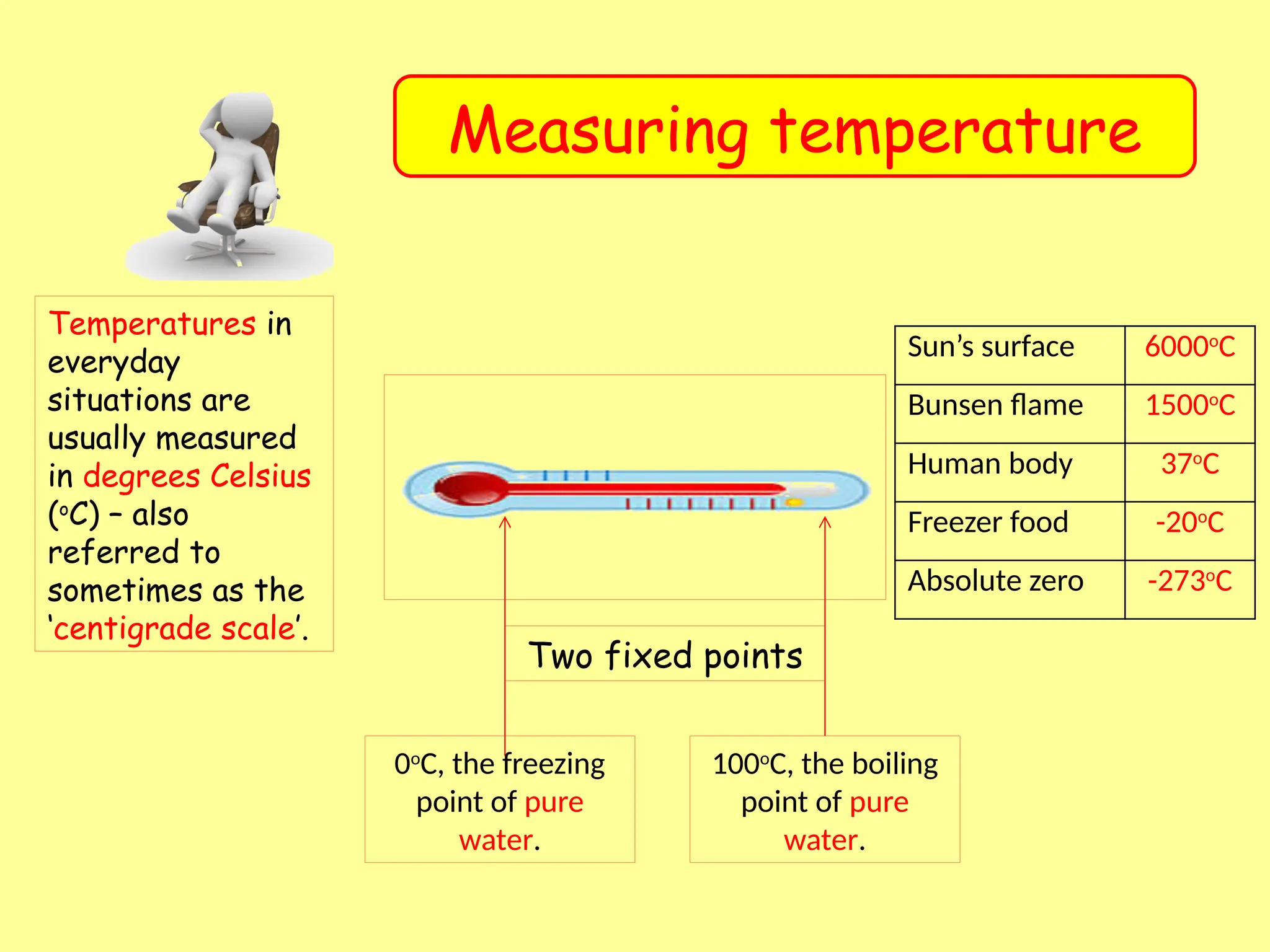
The freezing point in centigrade, also known as the Celsius scale, is the temperature at which a liquid turns into a solid. Water, for case, freezes at 0 degrees centigrade (0 C). This scale is wide used in scientific and everyday contexts due to its simplicity and practicality. The centigrade scale is establish on the freezing and boiling points of h2o, get it a commodious reference for many applications.
Measurement of Freezing Point
Measuring the freezing point in centigrade involves various methods, each suited to different types of substances and levels of precision. Here are some mutual techniques:
- Thermometer Method: This is the simplest method, affect the use of a thermometer to measure the temperature at which a heart begins to freeze. The thermometer is pose in the liquid, and the temperature is read as the liquid starts to solidify.
- Differential Scanning Calorimetry (DSC): This method uses a calorimeter to mensurate the heat flow associated with phase transitions. It provides precise measurements of the freezing point and is unremarkably used in research and industrial settings.
- Cryoscopy: This technique involves measuring the freeze point depression of a result. By bestow a known amount of solute to a solvent, the freezing point of the solvent is lour. The difference between the freeze points of the pure solvent and the solution can be used to influence the freezing point of the pure substance.
Importance of Freezing Point in Centigrade
The freezing point in centigrade is a critical argument in assorted fields, including chemistry, physics, engineering, and food science. Understanding the freeze point helps in:
- Material Science: The freeze point is crucial for understanding the demeanor of materials under different temperature conditions. This cognition is essential for developing new materials and amend live ones.
- Food Preservation: In the food industry, the freezing point is used to mold the optimum storage conditions for perishable items. Freezing food at the correct temperature helps preserve its quality and extend its shelf life.
- Chemical Processes: Many chemic reactions are temperature sensible, and knowing the freeze point of reactants and products is indispensable for contain these reactions. This ensures the desired outcomes and minimizes waste.
- Environmental Science: The freezing point of water is a key factor in translate climate patterns and weather phenomena. for case, the freeze point of water in clouds affects precipitation and conditions omen.
Applications of Freezing Point in Centigrade
The freezing point in centigrade has numerous applications across diverse industries. Some of the most significant applications include:
- Refrigeration and Air Conditioning: Understanding the freeze point of refrigerants is crucial for design effective cooling systems. The freeze point helps in selecting the right refrigerating and optimizing the scheme s execution.
- Pharmaceuticals: In the pharmaceutic industry, the freezing point is used to determine the stability of drugs and their storage conditions. Many drugs are sensible to temperature changes, and cognize the freeze point helps in keep their efficacy.
- Agriculture: The freezing point of h2o is all-important for understanding the impact of frost on crops. Farmers use this information to protect their crops from frost damage and secure a successful harvest.
- Geology: The freeze point of h2o is a key divisor in understanding geological processes such as glacier formation and soil freezing. This cognition is essential for canvass climate change and its impact on the Earth s surface.
Freezing Points of Common Substances
Here is a table of the freezing points in centigrade for some common substances:
| Substance | Freezing Point in Centigrade (C) |
|---|---|
| Water | 0 |
| Ethanol | 114. 1 |
| Mercury | 38. 83 |
| Gold | 1064. 18 |
| Silver | 961. 78 |
Note: The freezing points listed above are approximate values and can vary somewhat depending on the purity of the substance and environmental conditions.
Factors Affecting Freezing Point
Several factors can influence the freezing point of a substance. Understanding these factors is essential for accurate measurements and applications. Some of the key factors include:
- Purity: Impurities in a substance can lower its freeze point. This phenomenon is known as freeze point slump and is commonly used in cryoscopy to determine the molecular weight of solutes.
- Pressure: Changes in pressing can affect the freezing point of a nub. for instance, the freeze point of h2o decreases with increasing press, which is why ice can form at temperatures below 0 C under eminent pressure.
- Rate of Cooling: The rate at which a nitty-gritty is cooled can also involve its freeze point. Rapid cooling can result in supercooling, where the liquid remains in a liquid state below its freeze point.
Supercooling and Its Implications
Supercooling is a phenomenon where a liquid is cooled below its freeze point without solidifying. This occurs because the liquid lacks nucleation sites, which are necessary for crystal formation. Supercooling has several implications:
- Cloud Formation: Supercooled water droplets in clouds can conduct to the formation of ice crystals, which are essential for precipitation.
- Food Preservation: Supercooling is used in the food industry to preserve perishable items without freezing them. This method helps sustain the texture and caliber of the food.
- Material Science: Supercooling is used to study the properties of materials and evolve new ones with unequalled characteristics.
Supercooling is a fascinating phenomenon that highlights the complexity of phase transitions and the importance of understanding the freeze point in centigrade.

Freezing Point Depression
Freezing point slump is the phenomenon where the freeze point of a solvent is lowered by the add-on of a solute. This principle is wide used in various applications, include:
- Antifreeze: Antifreeze solutions, such as ethylene glycol in h2o, lower the freezing point of the coolant in vehicles, foreclose it from freezing in cold temperatures.
- De icing: De icing solutions, such as brine, are used to melt ice on roads and sidewalks by lowering the freeze point of water.
- Food Preservation: Adding salt or sugar to food can lower its freezing point, aid to preserve it by subdue bacterial growth.
Freezing point slump is a important concept in chemistry and has wide ranging applications in industry and everyday life.
Understanding the freeze point in centigrade is crucial for various scientific, industrial, and everyday applications. From material skill to food saving, the freeze point plays a critical role in many processes. By measuring and contain the freeze point, we can optimise these processes and develop new technologies. The freezing point in centigrade is a primal property that continues to be a subject of study and institution.
Related Terms:
- boil point fahrenheit
- boil point celsius
- is 3 degrees celsius freezing
- freezing point fahrenheit
- h2o freezes at what celsius
- freezing point kelvin