The Bohr Model of the atom, proposed by Niels Bohr in 1913, overturn our understanding of atomic structure. While the model is often discuss in the context of hydrogen and other uncomplicated atoms, it also provides worthful insights into more complex elements like oxygen. Understanding the Bohr Model Oxygen can help us grasp the fundamental properties and behaviors of this essential element.
Understanding the Bohr Model
The Bohr Model is a planetal model of the atom, where electrons orbit the nucleus in fixed, circular paths called orbits or shells. Each shell corresponds to a specific energy level, and electrons can jump between these levels by assimilate or emitting energy. This model is peculiarly utile for explaining the emission spectra of elements.
The Bohr Model of Oxygen
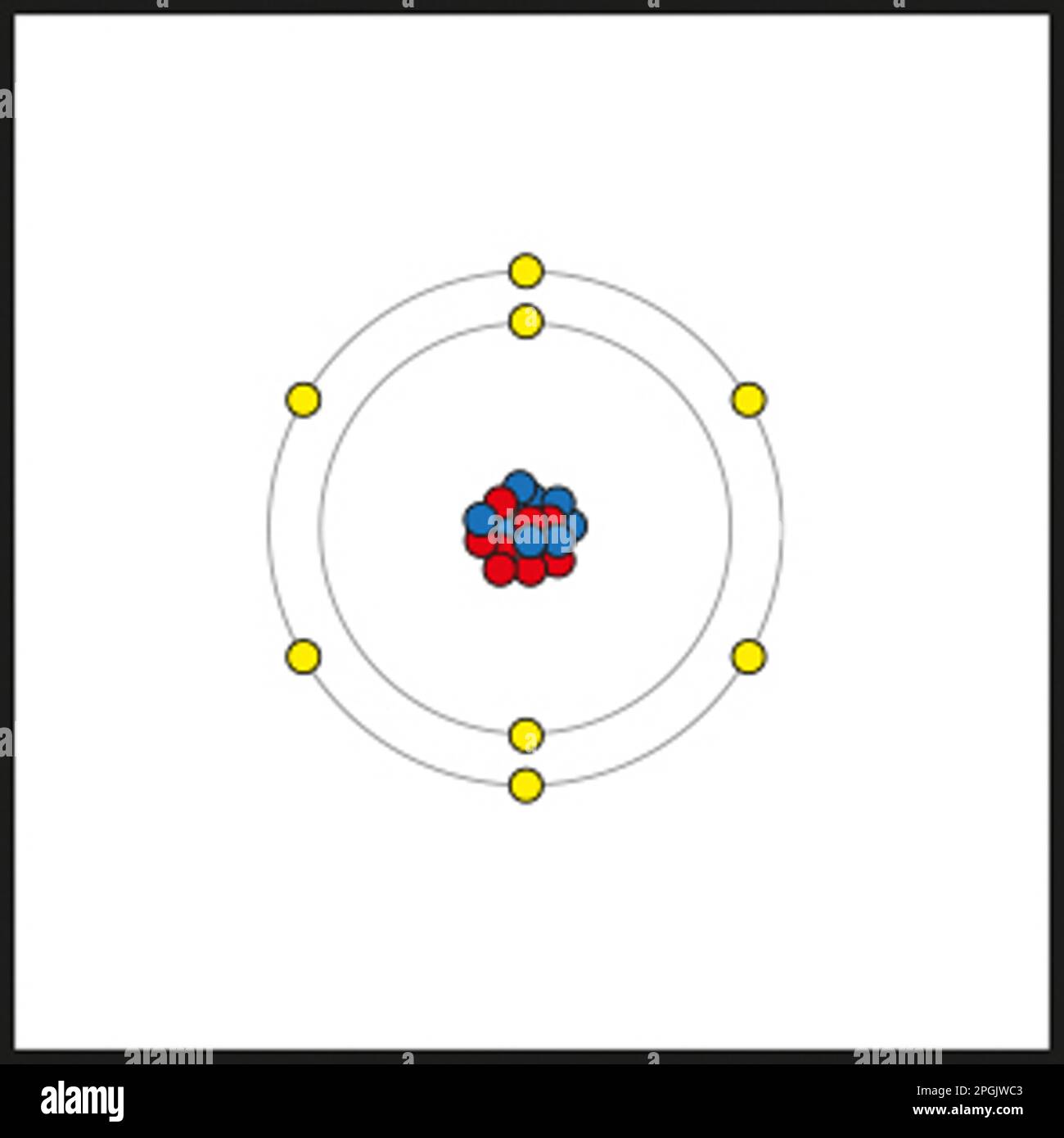
Oxygen, with the nuclear figure 8, has 8 protons and 8 electrons. In the Bohr Model Oxygen, these electrons are arrange in two main shells. The first shell, closest to the nucleus, can hold up to 2 electrons. The second shell, which is further out, can hold up to 8 electrons. Therefore, the electron configuration of oxygen in the Bohr Model is 2 electrons in the first shell and 6 electrons in the second shell.
Here is a uncomplicated representation of the Bohr Model Oxygen:
| Shell | Electrons |
|---|---|
| First Shell (K) | 2 |
| Second Shell (L) | 6 |
This configuration helps excuse why oxygen is extremely responsive. The second shell is not full occupy, which makes oxygen bore to gain or partake electrons to achieve a stable configuration.
Electron Configuration and Chemical Properties
The electron conformation of oxygen, as report by the Bohr Model, straight influences its chemical properties. Oxygen tends to form covalent bonds with other elements to complete its outer shell. for instance, in h2o (H 2 O), oxygen forms two covalent bonds with hydrogen atoms, sharing electrons to achieve a stable configuration.
Oxygen can also form ionic bonds. In compounds like sodium oxide (Na 2 O), oxygen gains two electrons from sodium atoms, forming a stable octet and resulting in a negatively charged oxide ion (O2 ).
Energy Levels and Spectra
The Bohr Model also helps explicate the emanation and absorption spectra of oxygen. When oxygen atoms absorb energy, electrons can jump to higher energy levels. As these electrons retrovert to their original levels, they emit energy in the form of photons. The specific wavelengths of these photons correspond to the energy differences between the levels, result in characteristic ghostlike lines.
for case, when oxygen molecules are stir, they can emit light in the ultraviolet and seeable regions of the spectrum. This is why oxygen's front can be detected through spectroscopical analysis, which is essential in fields like astronomy and environmental science.
Limitations of the Bohr Model
While the Bohr Model provides a useful framework for translate nuclear structure, it has several limitations. One of the main drawbacks is that it does not accurately describe the behavior of electrons in atoms with more than one electron. For elements like oxygen, the interactions between electrons are complex and cannot be full explained by the simple orbital model.
Additionally, the Bohr Model does not account for the wave like properties of electrons, which are better line by quantum mechanics. In more advanced models, such as the Schrödinger equating, electrons are draw by wave functions that provide a probability distribution of their positions.
Despite these limitations, the Bohr Model remains a worthful tool for introductory studies of atomic structure and for realize the canonical properties of elements like oxygen.
Note: The Bohr Model is a foundational concept in chemistry and physics, but it is crucial to discern its limitations when analyze more complex systems.
Applications of the Bohr Model
The Bohr Model has legion applications in assorted fields of science and engineering. Understanding the Bohr Model Oxygen can facilitate in the following areas:
- Chemical Bonding: The model helps explain how oxygen forms bonds with other elements, which is essential in understanding chemic reactions and the properties of compounds.
- Spectroscopy: The Bohr Model is used to interpret the emission and absorption spectra of elements, which is essential in fields like astronomy and materials skill.
- Education: The model serves as a fundamental concept in teaching nuclear structure and chemistry, supply a open and intuitive understanding of electron behavior.
By applying the Bohr Model to oxygen, scientists and educators can gain insights into the element's behavior and properties, which are essential for various scientific and technical advancements.

to summarize, the Bohr Model provides a fundamental understand of nuclear structure, include the Bohr Model Oxygen. By examining the electron configuration and energy levels of oxygen, we can excuse its chemic properties and doings. While the model has limitations, it remains a valuable tool for introductory studies and practical applications in several scientific fields. Understanding the Bohr Model Oxygen helps us appreciate the complexity and beauty of atomic structure and its role in the natural universe.
Related Terms:
- oxygen bohr rutherford diagram
- bohr model carbon
- bohr rutherford diagram for be
- bohr model neon
- bohr model chart
- oxygen bohr model explain